Rare earth elements are a collective term for 17 special elements, including lanthanide elements in the periodic table of chemical elements - lanthanide (La), cerium (Ce), praseodymium (Pr), neodymium (Nd), promethium (Pm), samarium (Sm), europium (Eu), gadolinium (Gd), terbium (Tb), dysprosium (Dy), holmium (Ho), erbium (Er), thulium (Tm), ytterbium (Yb), lutetium (Lu), as well as 15 closely related elements of the lanthanide system - yttrium (Y) and scandium There are 17 elements in Sc, called rare earth elements. Rare earths are important strategic resources, known as "industrial vitamins", and have been widely used in fields such as electronics, petrochemical, metallurgy, machinery, energy, light industry, environmental protection, agriculture, etc. Today, let's first introduce the first ranked metal lanthanum.
1、 Introduction to Lanthanum
In 1839, Swedish chemist Carl Mosander discovered "lanthanum" (named after the Greek word for "hidden"). Lanthanum is a metallic rare earth element with atomic number 57 and atomic weight 138.9055. Silver gray luster, soft texture, density 6.174g/cm ³, Melting point 921 ℃, boiling point 3457 ℃; The content of lanthanum in the crust is 0.00183%, second only to cerium in rare earth elements. Lanthanum has two natural isotopes: lanthanum 139 and radioactive lanthanum 138.
Lanthanum metal is a silver white metal that is soft and easy to cut. Lanthanum metal has active chemical properties and is easily soluble in dilute acids. It is easy to oxidize in the air, and fresh surfaces quickly darken when exposed to air. Lanthanum metal is generally stored in mineral oil or rare gases. Heating can burn, producing oxides and nitrides. Heating in hydrogen gas generates hydrides, which react strongly in hot water and release hydrogen gas. Lanthanum exists in monazite sand and bastnaesite. Lanthanum is a malleable and malleable silver white metal that can be cut open with a knife due to its soft texture; Melting point 921 ° C, boiling point 3457 ° C, density 6.174 grams/cubic centimeter. Lanthanum has active chemical properties and slowly corrodes in cold water, accelerating in hot water; Lanthanum can directly react with carbon, nitrogen, boron, selenium, silicon, phosphorus, sulfur, halogens, etc; Lanthanum compounds exhibit diamagnetism.
2、 Application of Lanthanum Metal
(1) Steel modifier
Adding lanthanum or mixed rare earth metals to steel can remove sulfur and oxygen, refine grain size, form microalloys, change the morphology and distribution of inclusions, reduce hydrogen diffusion coefficient, and improve resistance to hydrogen embrittlement and stress corrosion; Adding iron can purify molten iron, change graphite morphology, and prevent impurities from damaging spheroidization. Due to the widespread application of steel in various fields, lanthanum metal plays an important role in the development of high-performance products such as steel and cast iron.
(2) Reducing agent
Metal lanthanum and cerium have similar properties. Lanthanum chips are mixed with samarium oxide and compressed into blocks, and reduction reactions occur at high temperatures. High vapor pressure metals such as samarium can be obtained by vacuum distillation and purification using vapor pressure difference; The equipment for this process is a vacuum induction furnace or vacuum resistance furnace, where the reduction and distillation processes are carried out simultaneously, resulting in a simple process and minimal pollution.
(3) Metal square bar lining
Pure rare earth metals, due to their active chemical properties, are prone to react with oxygen, sulfur, and nitrogen to form stable compounds. When subjected to intense friction and impact, sparks can ignite flammable substances. Therefore, as early as 1908, it was made into flint. It has been found that among the 17 rare earth elements, six elements, cerium, lanthanum, neodymium, praseodymium, samarium, and yttrium, have particularly good arson performance. And lanthanum has the lowest price. People have made various incendiary weapons based on the arson properties of rare earth metals. For example, the 227kg American "Mark 82" missile uses rare earth metal liners, which not only produce explosive killing effects but also arson effects. The US air-to-ground "damping man" rocket warhead is equipped with 108 rare earth metal square rods as liners, replacing some prefabricated fragments. Static explosion tests have shown that its ability to ignite aviation fuel is 44% higher than that of unlined ones.
(4) Metal lanthanum wire foil
The metal lanthanum wire can absorb harmful gases such as oxygen, nitrogen, carbon monoxide, carbon dioxide, and water vapor released by the electron tube electrode due to bombardment and thermal diffusion, thereby maintaining the high vacuum of the electron tube. Various rare earth metals and alloy foils have a large neutron absorption area and can effectively capture neutrons. The wire and foil of lanthanum metal are widely used in fields such as electronics, lighting, and nuclear industry.
(5) Igniting alloy
In the early days of our country, various ignition alloys were made from mixed rare earth metals (RE, containing La25%) and Fe, and can be divided into military and civilian types. Military ignition alloys are made of RE60-80% (including La25%), Fe20-40%, and a small amount of Al, Ca, Si, and C, mainly used for manufacturing bullets, shells, and bombs, as well as ignition devices. Civil ignition alloys are made of RE75-80% (including La25%), Fe15-18%, and a small amount of Mg, Zn, Cu, Al, etc., with a ignition rate of ≥ 85%. They are mainly used for igniting flints in lighters and various toys. In addition, igniting alloys are also used for industrial steam lamps, welding torch igniters, and torch igniters.
(6) For non-ferrous metals
Metal lanthanum powder has a larger specific surface area, stronger activity, and better dispersibility than metal lanthanum blocks, and its applications in precision alloys, special metals, and catalysts are increasing day by day. Tungsten alloys, molybdenum alloys, and titanium alloys have problems such as low grain boundary strength and low temperature brittleness; During its processing, rare earth metal powders such as lanthanum powder are added and thoroughly mixed, which can utilize the effect of rare earth microalloying to effectively refine the structure, capture harmful elements such as hydrogen, and improve alloy performance.
(7) Metallic lanthanum target
Metal lanthanum targets are mainly used in fields such as coating and polishing. The use of thorium tungsten material as a thermionic cathode poses a radioactive problem, while lanthanum molybdenum cathode does not. Its emission performance largely depends on the surface active material layer of the material. Hao Shiming et al. used molybdenum as the substrate and lanthanum as the target material to prepare uniformly distributed lanthanum oxide thin films using pulse laser technology, and obtained excellent performance lanthanum molybdenum cathodes. CVD diamond film has good thermal conductivity and transparency, and is widely used, but the surface grain size and roughness of the film are large, resulting in low performance. By utilizing the reaction diffusion of carbon elements and rare earth metals on the surface of diamond to achieve surface polishing, the polishing speed can be accelerated and the precision of diamond films can be improved. High purity lanthanum targets have fewer impurities and vacancies, resulting in a more uniform structure and stable performance of the sputtered film layer.
3、 Application of Lanthanum Compounds (1) Lanthanum Oxide (La2O3)
White amorphous powder with a density of 6.51. Melting point 2315 ℃. Boiling point 4200 ℃. Slightly soluble in water, soluble in acids to form corresponding salts. Exposed to the air, it absorbs carbon dioxide and gradually transforms into lanthanum carbonate. Used for making special alloys, optical glass, etc. Extracted from lanthanum phosphate cerium ore or obtained by burning lanthanum carbonate or nitrate. Manufacturing various alloy materials, such as aluminum alloys for aircraft; Luminous materials, road signs and pavement luminous stones; Optical glass, such as cameras and camera lenses; Optical fiber; Advanced capacitors; Glass decolorizing agent to increase strength; Laser materials; Pigments and gloss agents for porcelain; Magnetoresistive material; Hydrogen storage materials; Mobile phone battery; Catalysts for organic chemical products, such as light conversion agricultural films.
(2) Lanthanum sulfate (La2 (SO4) 3)
Slightly soluble in cold water, with an increase in temperature and a decrease in solubility, insoluble in acetone. It is the least soluble rare earth metal sulfate. The most common is its octahydrate hydrated lanthanum sulfate, which is a colorless hexagonal crystal. It dehydrates at 500 ℃ and generates basic salts at 700 ℃. It can form various sulfate complex salts with alkali metal sulfates. La (HSO4) 3 is generated in sulfuric acid, and sulfides are generated when heated under the flow of sulfurized hydrogen gas. Hydrated lanthanum sulfate can be used for determining the atomic weight of elements, spectral analysis, and as a preservative and reagent.
8 hydrated lanthanum sulfate
(3) Lanthanum carbonate (La ₂ (CO Å))
Lanthanum carbonate can reduce the blood calcium and phosphorus levels in patients with high calcium and high phosphorus MHD, and is safe. It can be used to treat hyperphosphatemia accompanying dialysis in kidney disease patients.
Lanthanum carbonate, as a new type of phosphorus binder that does not contain aluminum or calcium, is gradually being used in the clinical treatment of hyperphosphatemia. Lanthanum carbonate has good phosphorus binding ability in acidic environments, and trivalent lanthanum ions are highly compatible with phosphorus in stomach acidic environments. They can tightly bind to phosphorus in food, forming insoluble and indigestible lanthanum phosphate, which is excreted with feces and can reduce blood phosphorus levels. There are no toxic side effects caused by increased intake of aluminum and calcium.
(4) Lanthanum hexaborate (LaB6)
It is an excellent electron emission material with high melting point (>2500 ℃), low vapor pressure, and low work function. Its electron emission performance is better than tungsten, and it has been widely used as an electron gun in electron microscopes, televisions, and cathode ray tubes.
LaB6 has a relatively low escape work and is an excellent cathode material, especially suitable for high-temperature and high current density devices. Based on its unique structure, it has excellent electronic activity. During heating, the metal La atoms diffused from the crystal cell can immediately supplement the metal La atoms evaporated from the surface, maintaining good cathodic activity on the LaB6 surface. Due to its high conductivity, good thermal and chemical stability, low escape work, and excellent cathode surface activity, LaB6 has been widely used in cathode emission and has become a good hot cathode material and field emission cathode material.
Crystal Structure Model of LaB6
(5) Lanthanum bromide oxide (LaBrO)
It has strong absorption characteristics for X-rays and can effectively convert X-rays into visible light. Using it to make medical X-ray fluorescence intensifying screens greatly improves imaging clarity compared to traditional calcium tungstate (CaWO4) intensifying screens, and reduces X-ray irradiation dose. It is especially suitable for fluoroscopy of sensitive areas of the brain and children and pregnant women
(6) Lanthanum bromide (LaBr3)
Light gray white powder, easy to absorb moisture; Density (25/4 ℃) 5.063g/mL; Melting point 7834 ℃; Boiling point 15775 ℃; Soluble. Cerium doped lanthanum bromide single crystal (LaBr3: Ce3+) is an excellent scintillator material with superior scintillation performance compared to sodium iodide, cerium doped lanthanum chloride, etc.
Lanthanum bromide LaBr3 (ce) scintillation crystal is the main component of the nuclear radiation probe. The scintillation probe can be used to detect X-rays and γ Ionizing radiation such as radiation. Lanthanum cerium bromide scintillation crystals have the advantages of high light yield, good energy resolution, short decay time, and small nonlinear response. They can be widely used in various fields such as international counter-terrorism, nuclear material control, safety inspection, energy, nuclear medicine, industrial metrology, and petroleum logging.
LaBr3 (ce) crystal of lanthanum bromide
(7) Lanthanum nitrate (La (NO3) 3)
White granular crystals, easy to wet dissolve; Boiling point 126 ℃; Melting point 40 ℃; Solubility: easily soluble in water, easily soluble in ethanol; Density: relative density (water=1) 2.05; Stability: Stable; Hazard sign 11); Main uses: used for making optical glass, fluorescent powder, ceramic capacitor additives, and petroleum refining processing catalysts
1. Used for the production of optical glass, steam lamp covers, fluorescent powder, and preservatives.
2. Ceramic capacitor additives and petroleum refining catalysts.
6 hydrated lanthanum nitrate

(8) Lanthanum chromate (LaCrO3)
Lanthanum chromate material is a composite oxide with a cubic perovskite structure at high temperatures (>1000 ℃), a theoretical density of 6.5g/cm3, and a melting point of 2490 ℃. Pure lanthanum chromate material is an intrinsic semiconductor, and after appropriate doping, it can become a p-type semiconductor with good conductivity. Due to the presence of oxygen in the chemical formula, the lanthanum chromate electric heating element prepared by using lanthanum chromate material as the substrate and adding other materials has good high-temperature oxidation resistance. The surface temperature can reach 1900 ℃ in an air environment, and the long-term stable working temperature in the furnace can reach 1700 ℃. Resistance heating elements used in high-temperature oxidation atmosphere electric furnaces; It consumes less energy and can accurately control temperature. It can be used for a long time in an oxidizing atmosphere and is suitable for high-precision automatic temperature control. Its furnace temperature stability can be within 1 ℃.
(9) Lanthanum silicate (La3Ga5SiO14)
It is an ideal material for producing high stability, high frequency, large bandwidth, low insertion loss, and small volume SAW filters. The La-Ca-Mn-O material, a type of perovskite manganese based oxide, exhibits giant magnetoresistance (CMR) effect, which has promoted the development of an emerging discipline - spin electronics, and has begun to be applied in many new electronic devices.
Gallium lanthanum silicate (LGS) crystal
Gallium lanthanum silicate (LGS) crystals, like quartz crystals, belong to the tripartite crystal system, but their electromechanical coupling coefficient is 2-3 times higher than that of quartz crystals. They are non deliquescent, insoluble in acids and bases, have zero temperature coefficient tangential, low SAW propagation rate, no phase transition, and do not require polarization treatment. They are excellent piezoelectric crystal materials. However, due to the fact that half of its components weigh Ga2O3, the high cost makes it difficult to compete with large and affordable quartz crystals. Therefore, devices made of LGS crystals are only used in a few fields that require excellent properties regardless of cost, such as aviation, aerospace, or military.
In recent years, due to the development of mobile communication, small intermediate frequency acoustic bulk wave filters made by LGS have been used in broadband wavelength division multiplexing (W-CDMA) systems and have once again received attention.
4、 The application range of lanthanum alloys
(1) Nuclear radiation shielding
Application principle: Using 1% boron and 5% rare earth elements gadolinium, samarium, and lanthanum, a 600mm thick radiation resistant concrete is made to shield the fission neutron source of a swimming pool reactor.
France has developed a rare earth radiation resistant material by adding borides, rare earth compounds, or rare earth alloys to graphite as the substrate. The filler of this composite shielding material is required to be evenly distributed and made into prefabricated parts, which are placed around the reactor channel according to the different requirements of the shielding area.
(2) Hydrogen storage materials
Energy is the foundation of national economy and scientific and technological development. The development and utilization of green and efficient hydrogen energy can effectively alleviate the energy crisis, and the storage and transportation of hydrogen energy are key technologies. Metal alloy solid hydrogen storage has the advantages of high energy density, safety and environmental protection. The LaNi5 alloy discovered in 1970 is an excellent hydrogen storage material that can store approximately 160 liters of hydrogen per kilogram, reducing the volume of high-pressure hydrogen storage cylinders to 1/4. By utilizing its ability to "breathe" hydrogen gas, hydrogen gas with a purity of 99.999% can be purified to 99.9999%, and can also be used as a catalyst for hydrogenation or dehydrogenation reactions in organic synthesis. By utilizing its ability to absorb hydrogen, release heat, and exhale hydrogen, heat can be transferred from low to high temperatures, and used to make "heat pumps" or "magnetic refrigerators". Yan Huizhong et al. studied the structure, hydrogen storage performance, electrochemical performance, treatment process, and application of lanthanum containing binary and multivariate hydrogen storage alloys, focusing on how to improve the hydrogen storage capacity of materials. Currently, the industrialization technology is relatively mature. Research has shown that the purity of raw materials affects the microstructure of materials, and high-purity raw materials can effectively improve the hydrogen storage capacity and service life of hydrogen storage alloys.
At present, the biggest use of this hydrogen storage material is as a negative electrode material for rare earth nickel hydrogen batteries. Rare earth nickel hydrogen batteries have great similarities and substitutability with nickel cadmium batteries in terms of structure, performance, and specifications, but they do not contain toxic elements such as cadmium and mercury. The batteries have high capacity, good consistency, wide temperature range, long lifespan, and can be recharged and discharged more than 500 times, making them environmentally friendly and green batteries. In order to reduce costs, this hydrogen storage alloy often uses lanthanum rich mixed metals with La ≥ 40% as raw materials. Rare earth nickel hydrogen batteries are currently widely used in portable computers, office equipment, and power tools. The most promising development is the power battery used in automobiles and motorcycles.
A material used as an anode material for nickel hydrogen batteries is La (Ni3.6Mn0.4Al0.3Co0.7). Due to the high cost of extracting other lanthanide elements, mixed rare earths containing over 50% lanthanum are used instead of pure lanthanum. This compound is an AB5 type intermetallic compound.
(3) Magnetic cooling material
Magnetic refrigeration refers to a new refrigeration technology using magnetic materials as the medium. Its basic principle is to use the magnetocaloric effect of magnetic cooling materials (i.e., the magnetic cooling material releases heat to the outside world during isothermal magnetization, and absorbs heat from the outside world during adiabatic demagnetization) to achieve the purpose of refrigeration. The magnetic cooling medium must have a giant magnetic entropy change. La-Fe series compounds have a NaZn13 structure, and there is a giant magnetic entropy change within the phase transition temperature range. Adding an appropriate amount of other elements to La-Fe series compounds can effectively increase their Curie temperature and achieve excellent magnetic cooling effects. They are currently the most promising room temperature magnetic cooling materials for practical use, but their binary alloys are unstable. The research focus at home and abroad is on the preparation process of this series of alloys.
(4) Shielding coating
Electromagnetic radiation is an important source of pollution in the information society, and shielding is one of the most effective methods to resist electromagnetic interference. Lanthanide based electromagnetic shielding coatings have high shielding efficiency for electromagnetic waves, but their impedance is higher than that of silver series. Adding rare earths can adjust their electromagnetic parameters, reduce impedance, and improve shielding performance. Adding lanthanum to the lanthanide based electromagnetic shielding coating to prepare Cu La based coating improved the conductivity and electromagnetic shielding performance of the coating. Its electromagnetic shielding effectiveness reached 89dB for electromagnetic waves ranging from 30MHz to 1.5GHz, indicating good shielding effectiveness.
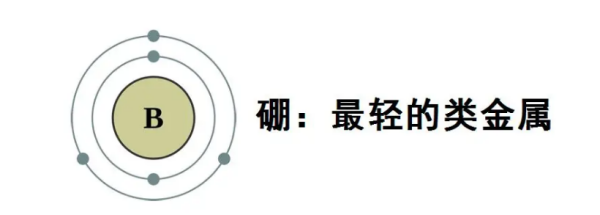
Boron is a chemical element with the chemical symbol B and atomic number 5, and is a type of metal. Boron is not produced by the synthesis of stellar nuclei, and almost all boron is produced by cosmic ray spallation and supernova explosions. Boron is a low abundance element in both the solar system and the Earth's crust. On Earth, boron generally exists in the form of borate.
1. Characteristics
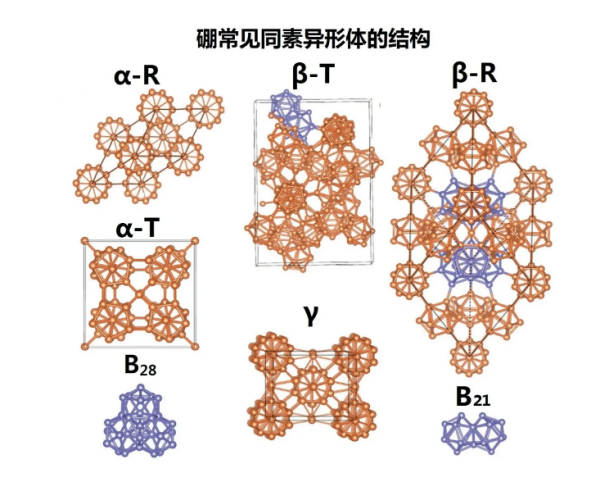
① Allotrope
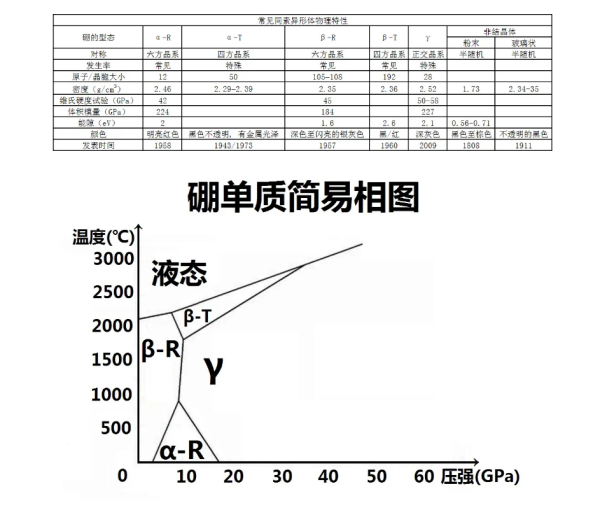
Boron has various allotropes, including crystals and amorphous forms. Crystal boron can form stable covalent bonding molecular networks similar to carbon. The commonly known crystalline boron is α- Rhombic boron( α- R) β- Rhombic boron( β- R) And β- Tetragonal boron( β- T) ; In addition, in certain specific situations, α- Tetragonal boron( α- T) γ Orthorhombic boron and other allotrope structures can also be prepared. There are two known forms of amorphous crystals, one is fine powder and the other is a glassy solid. Although at least 14 or more boron allotropes have been published, these discovered compositions are based on insufficient evidence, have not been experimentally confirmed, are considered mixed allotropes, or contain impurities to stabilize their boron structure. And boron β- The presence of diamond shaped boron crystals is the most stable, followed by the others, resulting in a low conversion rate at room temperature, and therefore the five different forms can coexist at room temperature. Amorphous powdered boron and polycrystalline boron β- Diamond boron is the most similar, with the latter being a very hard gray black material that is ten percentage points lighter than aluminum and has a melting point (2080 ° C) several hundred degrees higher than steel.
Elemental boron can be found in stardust and meteorites, but it does not exist in environments with high oxygen content on Earth. Because it is not easily extracted from its compounds. Previous extraction methods included reducing boron trioxide to a metal such as magnesium or aluminum. However, the product of this method is easily mixed with other metal borides. Currently, boron purification involves reducing volatile boron halides with hydrogen at high temperatures. The semiconductor industry requires the preparation of very pure boron, mainly through zone melting or Chaikelasky decomposition of ethyl boron at high temperatures. Preparing boron single crystals from pure boron is more difficult due to polymorphism and boron tendency to react with impurities; The typical crystal size of boron is about 0.1mm.
α- Diamond boron:
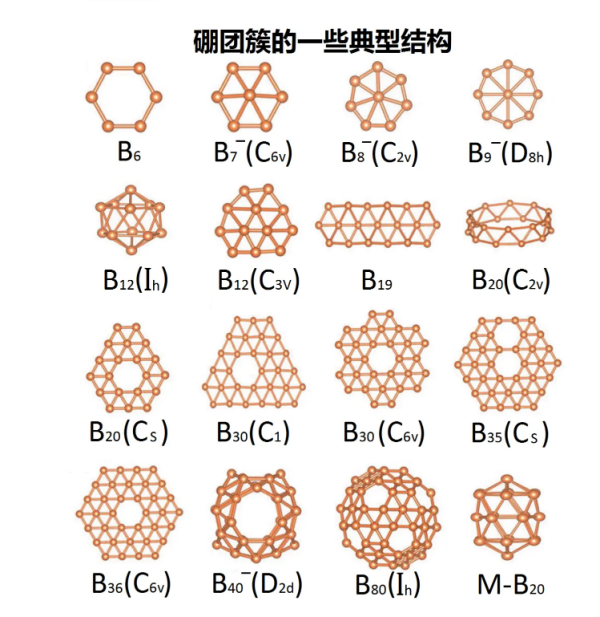
α A rhombohedral boron crystal is a unit cell containing 12 boron atoms. The unicellular structure of these B ₁ ₂ is a regular icosahedral structure where each unicellular body binds to five adjacent unicellular bodies. If these bonds are conventional covalent bonds, then each boron contributes five electrons. However, boron only has three valence electrons, so scientists believe that the B ₁ ₂ icosahedron is formed by the lack of bonding among three central electrons, meaning that electronic charges accumulate at the center of a triangle formed by three adjacent atoms.
A single B ₁ ₂ regular icosahedron is unstable; Although boron is not a molecular compound, it contains unconventional covalent bonds.
α- Tetragonal boron:
Pure α The synthesis of tetragonal boron crystals can only be carried out on the substrate plate of isotropic boron carbide (B Å ₀ C ₂) or nitride (B Å ₀ N ₂), α The tetragonal boron crystals are deposited on a layered substrate and form a thin film. And most of them α The synthesis of tetragonal boron crystals is carried out in a mixture of boron carbides or nitrides.
β- Diamond boron:
β A diamond shaped boron crystal is a single cell containing 105-108 boron atoms. The majority of atoms consist of B ₁ ₂ discrete icosahedra; An icosahedron with a few intersecting components, including two triangular polyhedral B ₁₀ units and a central boron atom. Previously, researchers did not understand the importance of α or β Which of the two structures is more stable, but later a consensus was gradually reached on this, β- Diamond boron is the most stable boron allotrope in thermodynamics.
β- Tetragonal boron:
In 1960, scientists synthesized BBr Å by hydrogen reduction on hot tungsten, rhenium, or tantalum wires in an environment of 1270 to 1550 ° C Celsius through chemical vapor deposition β Tetragonal boron crystal. Subsequent research further conducted this synthesis and confirmed that there were no impurities in the product. Reducing with metal not only produces metal boride impurities, but also causes β Tetragonal boron coexists with other phases.
γ Boron:
this γ- The state is a B ₁ ₂ icosahedron arranged in crystals similar to sodium chloride, and two forms of atoms, B2. It can be generated by compressing different types of boron to 12-20GPa and heating to 1500-1800 ° C, and maintaining stability in the environment. Evidence suggests that there is a significant transfer of charge from the B ₂ atomic pair to the B ₂ icosahedron in this structure, and this significant long-range electrostatic interaction can also be inferred from lattice dynamics.
In 1965, literature published the existence of boron in this state, but at this time, the structure and chemical composition of this type of boron were uncertain. Its structure was later confirmed by X-ray crystallography after being predicted and calculated through the "ab initio calculation method" of crystal structure.
Cubic boron:
In 1969 and 1976, literature was published on the observation of cubic boron allotropes in argon plasma experiments. This cube of boron contains 1705 ± 3 atoms with a density of 2.367g/cm ³, Belongs to unicellular crystals. Although this allotrope is occasionally mentioned in literature, there have been no subsequent official publications, research confirmations, or any evidence to deny its existence. In 1982, a new literature mentioned that the number of atoms in this single cell crystal indicates that it has a different structure from other common boron allotropes of regular icosahedra.
High voltage superconducting boron:
Pressurizing boron to over 160 GPa will result in an undefined phase state. Unlike other phases, this type of boron is not a semiconductor, but a metal and exhibits superconductivity. The critical temperature is 4K at 160GPa and rises to 11K at 250GPa. Under this pressure, the structure of boron will undergo a transformation, and theoretically, the original icosahedral structure will decompose. The current speculated structural state includes a face centered cubic structure similar to aluminum, α Gallium crystal structure and indium like body centered tetragonal structure. Like the transformation of iodine, the transformation of boron between non-metallic and metallic phases is believed to be caused by the closure of energy gaps, rather than just a simple structural transformation.
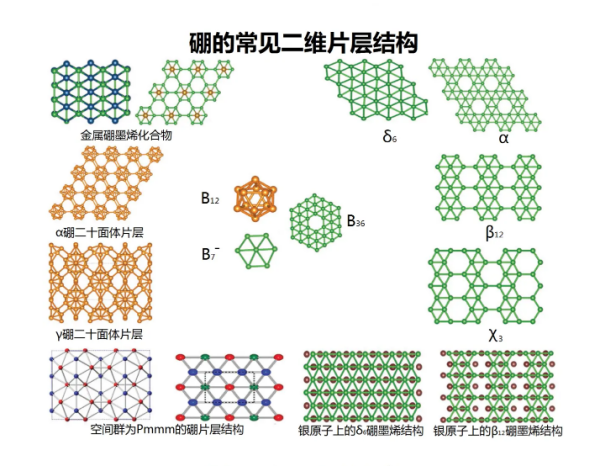
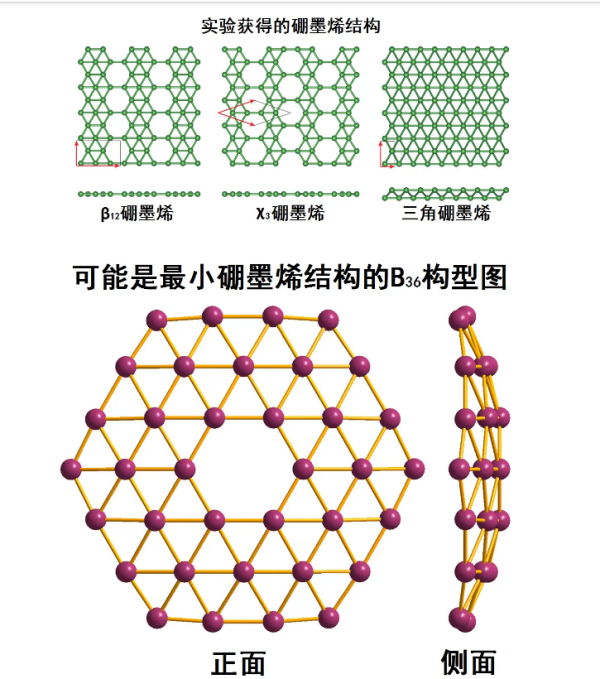
Borographene:
At present, it has been studied that boron can form several two-dimensional single-layer planar atomic structures similar to graphite in extreme cases. These two-dimensional structures are collectively referred to as borographene, and more structures have been predicted in theory waiting to be discovered. Borographene was first theoretically predicted to exist in the mid-1990s, and different borographene structures were experimentally confirmed in 2015.
Under high vacuum conditions, boron forms thin layered crystals and metal boron compounds on the metal surface. Their atomic structure is still controversial, but it is likely composed of a mixture of triangular and hexagonal structures, as shown in the following figure. Due to boron being an electron deficient element, it forms various interacting bonds in the multi center plane.
Boroglobulene:
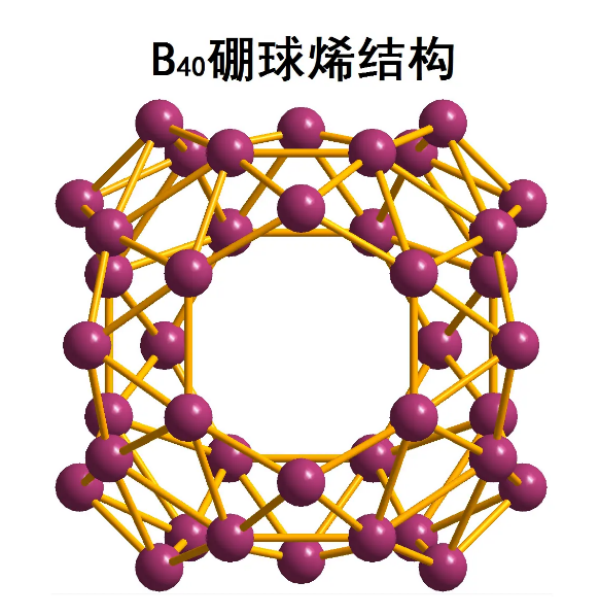
This spherical allotrope molecule, boroglobulene (B ₄₀), was published in July 2014.
B ₄₀ is a cluster molecule containing 40 boron atoms. Borospherene is similar to C ₆₀ in that it also has a spherical structure, but has different symmetries. The newly discovered boron spherene molecules also have an unusual heptagonal surface.
B ₄₀ has a unique axial symmetry (can rotate 180 °), but it is not as nearly spherical as C ₆₀ with icosahedral symmetry. It has a baseball like D ₂ d four corner anti prism symmetric structure. B ₄₀ has four heptagonal rings and two six unit rings, which together support 48 triangular boron atoms, forming four sets of 8 boron atomic structures and two sets of 4 boron atomic structures. Each boron atom binds to four to five other boron atoms. At first, there were two possible predictive structures for B ₄₀, namely sheet like structures and closed cage like structures. Spectral analysis shows that the experimentally synthesized B ₄₀ has a cage like structure, but these cage like structures are not very uniform. Some boron atoms will extend out, making the surface of B ₄₀ even smoother than Buck's sphere.
Amorphous boron:
Amorphous boron contains an icosahedral structure of B ₁ ₂, but randomly binds to each other in an unfixed order. Pure amorphous boron is generated by thermal decomposition of ethylboron below 1000 ° C. Annealing at 1000 ° C will transform amorphous boron into β- Diamond boron. Non crystalline boron nanowires (diameter 30-60nm) or fibers can be prepared by splash deposition and laser assisted chemical vapor deposition; Similarly, annealing at 1000 ° C can transform into β- Diamond boron nanowires.
② Boron compounds and their chemical properties
Boron is a relatively rare element, and due to the difficulty in preparing pure boron, there is relatively little research on elemental boron. The chemical properties of boron are more like silicon compared to aluminum. Crystal boron is generally inert in chemistry and can resist corrosion by hydrofluoric acid or hydrochloric acid at room temperature. Powdered boron can be slowly eroded by hot concentrated hydrogen peroxide, hot concentrated nitric acid, hot concentrated sulfuric acid, or a mixture of hot sulfuric acid and chromic acid.
The rate of boron oxidation depends on the crystallinity, particle size, purity, and temperature of boron. Boron does not react with air at room temperature, but it burns to form boron trioxide at high temperatures. Boron can also react with halogens to form trihalides, but in reality, purer trihalides are usually made from boron oxides.
Boron is a common compound with a+3 oxidation state, which can form compounds such as oxides, sulfides, nitrides, and halides.
Trihalides:
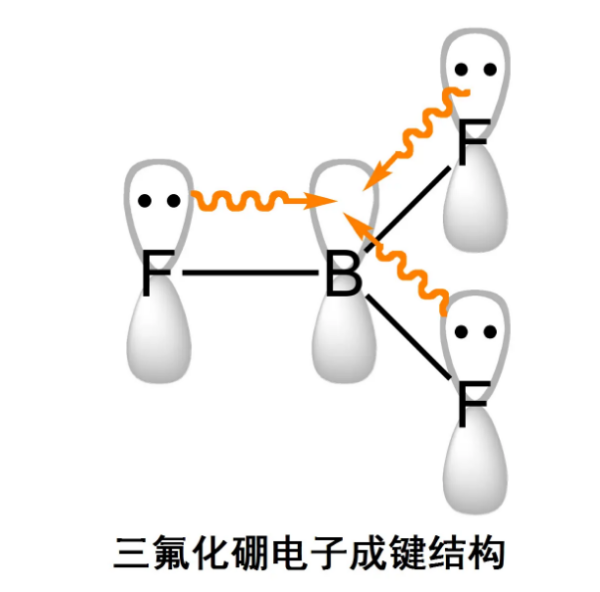
Trihalides adopt a planar triangular structure. Trihalides are Lewis acids that easily form adducts with electron pair donors as Lewis bases. For example, fluoride ions can combine with * * * to form tetrafluoroborate anions**** It can be used as a catalyst in the petrochemical industry. Trihalides react with water to form boric acid.
Unlike aluminum trihalides, boron trihalides are monomers. But the rapid halogen exchange reaction indicates that they can also undergo reversible dimerization reactions. But mixed halides are difficult to separate pure substances.
In * * * *, the boron atom is sp ² Hybridization, with a planar triangular structure and D3h symmetry group, is consistent with the prediction of valence layer electron pair repulsion theory. Although the B-F bond is a polar covalent bond, its molecular symmetry offsets the dipole moment, resulting in a dipole moment of 0**** Unlike carbonate ions, which are isoelectronic, * * * is an electron deficient compound.
****The B-F bond length (1.30 Å) in is shorter than the predicted single bond length, possibly due to the presence of large π bonds. Due to its planar structure, the empty p-orbitals of boron can be conjugated with the full p-orbitals of three fluorine atoms, resulting in electron delocalization and shorter bond lengths**** The electrons will be fed back to the ligand, causing the π orbital electrons of fluorine to delocalize into the p orbital of boron, forming the commonly known π 64 bond, which has a bond order of 1, resulting in a total bond electron number of 8.
Boron fire finished:
Boron is a compound of boron and hydrogen, with the general formula BxHy. These compounds are usually not present in nature. Boron is a type of electron deficient compound and therefore highly reactive to electron donors. Many boron can easily oxidize after coming into contact with air, while others can undergo severe oxidation. The parent material BH Å is known as ethylboron flame retardant, but it generally exists as a gaseous intermediate that forms a dimer of ethylboron flame retardant (B ₂ H ₆). Borons with higher molecular weights are generally composed of polyhedral boron clusters, and there are some isomers, with some cluster like compounds having coordination numbers greater than 4. For example, B ₂ ₀ H ₂ ₆ has two isomers formed by 10 atomic clusters.
The most important boron flame retardant is ethylboron flame retardant and its two cracking products, pentaboron flame retardant (9) and decaboron flame retardant (14). According to the ratio of boron to hydrogen, neutral boron fires can generally be divided into BnHn+4 types of low hydrogen boron fires and BnHn+6 types of multi hydrogen boron fires. In addition, there are a large number of boron fire anions, which can be commonly divided into three categories:
I: BnHn ² This type of ion bonding is similar to the corresponding neutral boron flame retardant molecule, with a polyhedral closed structure of intact boron atoms, such as [B ₁₂ H ₁₂] ² ⁻.
II: Neutral boron can undergo deprotonation or form anions with H ⁻, such as tetrahydroboron ions, BH ₄⁻.
III: Anions derived from boron hydride unit clusters, such as B ₁₈H ₂ ₁⁻.
Due to the lower electronegativity of boron compared to hydrogen, boron has a positive oxidation number during combustion. The average oxidation number of boron is the ratio of hydrogen to boron in the molecule. For example, in the completion of boron combustion, the boron oxidation state is+3, while in the completion of boron combustion B ₁₀₀₁₄, the boron oxidation state is 7/5. In these compounds, the oxidation state of boron is usually not an integer.
The naming of neutral boron is similar to organic alkanes, but the number of hydrogen atoms must be indicated in parentheses after it. When naming boron fire finished anions, the charge of the anions should be indicated in parentheses after the parent substance. If necessary, the number of hydrogen atoms can be indicated before the structural type. A prefix is required to indicate the structural type of complex boron flame retardant with isomers.
Bond formation after boron firing:
Boron is a compound that lacks electrons, meaning that there are not enough electrons to form a 2-center 2-electron bond between all adjacent atomic pairs in the molecule. Currently, boron with high molecular weight involves the following bonds:
Ⅰ: Three center dual electron hydrogen bridge
II: Three center double electron B-B-B bond
III: 2-center 2-electron bond (in B-B, B-H, and H-B-H)
The multi center bonding theory has expanded the study of boron bonding. For example, in the icosahedral ion [B ₁₂ H ₁₂] ² In ⁻, the fully symmetric molecular orbitals are uniformly distributed among 12 boron atoms. Theoretical chemistry continues to study the development theory of bond formation during boron combustion, such as Stone's tensor surface harmonic cluster bond theory. The current development direction is four center double electron bonds.
Boron flame retardant chemical properties:
The simplest type of boronic acid is methylboronic acid, which is a very strong Lewis acid. Although the molecules themselves are unstable and form dimers, the adducts formed by the reaction of methyl boron with tetrahydrofuran and dimethyl sulfoxide can be used for borohydride reactions. Other boron compounds are electron deficient and can react violently with reagents that provide electrons, forming electron free molecules. For example, ethylboron can react with alkali metal hydride to form alkali metal boron hydride after firing.
Low molecular weight boron will release a large amount of heat when ignited with air, and unless the reaction dose is very small, an explosion will occur. This is not caused by the unstable combustion of boron, but because the product boron trioxide is a solid.
The formation of solids in oxidation reactions releases additional energy. In contrast, many group boron fire anions, such as [B ₁₂ H ₁₂] ² ⁻ does not react with air; The salts of these anions are metastable because the group structure creates a very high activation energy barrier for oxidation.
When treated with a strong alkali, boron with higher molecular weight can undergo deprotonation. For example:
Boron with larger molecular weight can also be used as a weak acid. For example, pentylenethiocarbamate (9) reacts with trimethylphosphine.
This reaction can produce derivatives that are considered to be unknown superclustered boron compounds. Acidity increases with the number of boron atoms. The pKa of B ₁₀ H ₁₄ is 2.7.
The brief formation of methyl boron flame during dissociation from ethyl boron flame can react with higher molecular weight boron flame to obtain a linked boron flame composed of two small boron flame subunits shared by boron atoms.
The subunits in the nested boron flame are connected by a B-B bond and can be produced by ultraviolet irradiation of the nested boron flame. Using platinum dibromide as a catalyst, some B-B coupled boron catalysts can be prepared.
The reaction of boron with acetylene can generate carbon boron flame, and the icosahedral cluster carbon boron flame (C ₂ B ₁₀H ₁₂) is particularly stable.
Boron can act as a ligand in coordination compounds. The Hapto number is η¹ reach η The tactile sensation of ⁶ is obtained by bridging hydrogen atoms or obtaining electrons from boron boron bonds. For example, nested boron pyrotechnic (10) can replace ethylene in Zeiss salt to produce iron Fe( η²- B ₆ H ₁₀ (CO) ₄.
Boron fire extinguishing application:
The main chemical application of boron combustion is the hydroboration reaction. Generally, boron flame retardant adducts are used, such as boron flame retardant adducts with tetrahydrofuran or dimethyl sulfide. These adducts have similar effects to boron flame retardant and do not have the same risk of explosion as boron flame retardant.
Boron can also be used for neutron capture in cancer treatment. Commonly used are boron flame retardant derivatives of sulfur hydrides, such as Na ₂ [B ₁ ₂ H ₁ (SH)]. Due to the high neutron capture cross-sectional area of boron-10, boron based flame retardant derivatives are highly suitable for use in this type of treatment.
Borate:
Borates refer to anionic derivatives formed by bonding boron with oxygen. Borates can not only refer to tetrahedral boron oxide anions, but also refer to compounds derived from any boron oxygen bond. Borates with more atoms can be composed of triangular or tetrahedral structures, which can be connected together by sharing oxygen atoms and exhibit circular or linear structures. Boron usually exists in the form of borate in nature, such as borate and borosilicate.
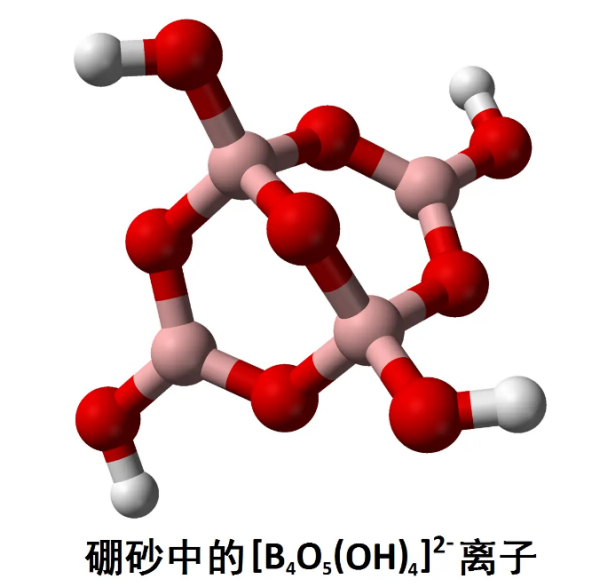
Borate structure:
The simplest borate anion is the orthoborate ion, [BO Å] ³ ⁻, many solid salts can be formed, such as calcium borate [Ca Å (BO Å) ₂]. Orthoborate ions have a planar triangular structure and are isoelectronic with carbonate ions, resulting in a similar structure. Boron is obtained by using sp ² Hybrid orbitals are used to form bonds. But compounds of orthoborate do not necessarily contain planar triangular ions, such as gadolinium orthoborate (GdBO Å), whose crystal structure contains triborate ions [B Å O ₉] ⁹⁻, which only decompose into planar [BO Å] at high temperatures ³ ⁻.
Acidity:
All borates can be considered derivatives of boric acid (H Å BO Å). Boric acid can serve as a Bronsted acid and is a weak proton donor (pKa approximately 9), but it is also a Lewis acid that can accept electrons. In water, it appears as a Lewis acid that receives electrons from the hydroxide ions produced by hydrolysis.
Therefore, boric acid is acidic because it interacts with hydroxide ions from water to form a tetrahydroxyborate complex [B (OH) ₄] ⁻, and releases the corresponding protons left by the proton self migration reaction of water molecules:
In the presence of cis glycols such as mannitol, glucose, sorbitol, and glycerol, pKa will decrease to around 4.
Polymer ion:
At a pH close to neutral, boric acid undergoes a condensation reaction, forming polyborate anions. The well-known polyborate anions include triborate anions (1-), tetraborate anions (2-), and pentaborate anions (1-). The condensation reaction of forming tetraborate anion (2-) is as follows:
The tetraborate anion structure consists of two tetrahedra and two triangular boron atoms symmetrically arranged in a polymerized double ring structure. These two tetrahedral boron atoms are connected together by an oxygen atom, and each tetrahedral boron atom also carries a negative charge, which is brought about by the hydroxyl groups connected to their sides. This complex ion exhibits three rings: two polymerized hexagonal boron rings and one twisted octagonal ring. Each ring is composed of a series of alternating boron and oxygen atoms. The borate ring is a very common structural basis in polyborate ions.
Tetraborate anions exist in mineral borax in the form of octahydrate Na ₂ [B ₄ O Å (OH) ₄] · 8H2O. The chemical formula of borax is usually represented by a more compact symbol, Na2B4O7 · 10H2O. High purity sodium tetraborate solution is commonly used as a standard solution for titration analysis.
Various metals can form borates. It is generally obtained by reacting metal oxides with boric acid or boron oxide. The following examples include linear chains of 2, 3, or 4 ternary boric acid structural units, each of which shares an oxygen atom with adjacent units:
Diborate [B ₂ O Å] Å ⁻, found in Mg ₂ B ₂ O Å (Suianshi)
Triborate [B Å O ₇] ⁻, found in CaAlB Å o ₇ (boracite)
Tetraborate [B ₄ O ₉] ⁶⁻, found in Li ₆ B ₄ O ₉
Metabolites such as LiBO ₂ contain a chain of ternary boric acid structural units, each of which shares two oxygen atoms with adjacent units, while NaBO ₂ and KBO ₂ contain cyclic [B Å O ₆] ³ ⁻ Ion.
Boric acid ester:
Boric acid esters are a class of organic compounds formed by the esterification reaction of boric acid with alcohols. There are two main types of borate esters: orthoborate esters [B (OR) Å] and metaborate esters, B Å O Å (OR) Å. Metaborate has a 6-membered boron oxide ring structure.
The reaction usually involves the addition of a dehydrating agent, such as concentrated sulfuric acid. Boric acid esters are volatile and can be purified by distillation. Esterification reaction can be used for borate analysis and boron content analysis in steel. Like most boron compounds, borate esters produce a unique green flame when burned. This property can be used for qualitative analysis of the presence of boron.

Boric acid esters spontaneously form after being treated with glycols such as sugars, and their reaction with mannitol is the basis for boric acid titration analysis.
Metaborate is a strong Lewis acid that can trigger epoxide polymerization reactions. The Lewis acidity of n-boric acid esters determined by the Gutmann Beckett method is relatively low.
Trimethyl borate [B (OCH Å) Å] is used to prepare precursor boric acid esters used in the Suzuki reaction. Asymmetric boric acid esters can be prepared by alkylation of trimethylboric acid esters:
These esters can be hydrolyzed to generate boric acid, which is mainly used in the Suzuki reaction.
Boride:
Borides are compounds formed by boron and low electronegativity elements, such as silicon borate (SiB Å and SiB ₆). Borides are a very large group of compounds that typically have a high melting point and are more similar in properties to covalent compounds compared to ionic compounds. Some borides exhibit very useful physical properties. Borides can also represent icosahedral borides such as B ₁ ₂ As ₂ (arsenic has higher electronegativity than boron). Boron in borides is in a negative oxidation state, while in some compounds it is in a non integer state.
Borides can be easily divided into boron rich borides and metal rich borides, such as the extreme yttrium borate (YB ₆₆) to the other extreme neodymium iron boron magnet Nd ₂ Fe ₁₄ B. The current default definition is as follows: if the ratio of boron atoms to metal atoms is 4:1 or higher, it belongs to boron rich borides; if it is lower than 4:1, it belongs to metal rich borides.
Boron rich borides:
The main group metals, lanthanide elements, and actinide elements can form various boron rich borides, with YB ₆₆₆ being the most abundant.
The properties of boron rich borides vary depending on the compound, and they can be used in semiconductors or superconductors. There may be diamagnetism, paramagnetism, ferromagnetism, or antiferromagnetism. They are mostly stable and have high melting points.
Some metals can form dodecahedral borides, while others (such as yttrium, zirconium, and uranium) can form borides with cubic octahedral boron atoms as the structural framework.
Lanthanum borate (LaB ₆) is an inert refractory material that can be used for chemical electrodes because of its low work function, which gives it a high electron thermal emissivity. YB ₆₆ crystal melted through indirect heating can be used as a monochromator for low energy X-ray synchrotron.
Rich metal borides:
Transition metals tend to form metal rich borides. Rich metal borides are also relatively inert and have a high melting point. Some metal rich borides are easily formed and can be used for manufacturing turbine blades, rocket nozzles, etc., such as AlB ₂ and TiB ₂. Recent research on such borides has shown that they have many unique properties, such as MgB ₂ being a superconductor at 39K, and the extreme incompressibility of OsB ₂ and ReB ₂.
Boride structure:
Boron rich borides contain a three-dimensional framework of polyhedra formed by boron atoms. Rich metal borides contain boron atomic units, such as B ₂ units, boron chains, or boron lamellar structures.
Examples of different types of borides are:
Independent boron atoms, such as Mn ₄ B
B ₂ units, such as VB ₂
Boron atomic chains, such as FeB
Boron layer, such as CrB ₂
A three-dimensional polyhedral boron skeleton, such as NaB ₁Å with an icosahedral boron skeleton
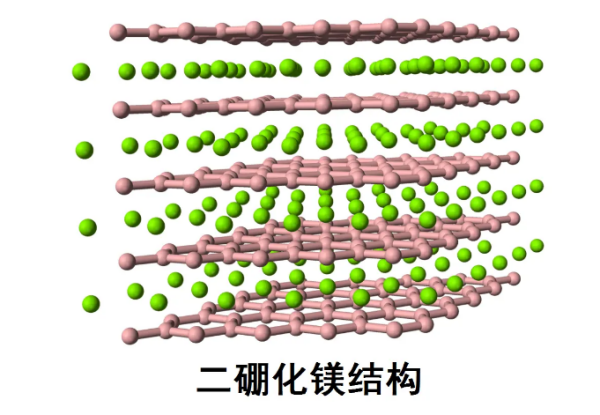
Boron nitride:
Boron nitride can form various structures similar to carbon allotropes, such as graphite, diamond, nanotubes, etc. The diamond-like boron nitride structure is called cubic boron nitride, where boron atoms replace the tetrahedral carbon atoms in the diamond. One out of every four B-N bonds can be regarded as a coordinate covalent bond, where the nitrogen atom serving as a Lewis base provides two electrons to the center of the boron atom serving as a Lewis acid. Cubic boron nitride can be used as an abrasive due to its hardness comparable to diamond. Graphite like boron nitride is called hexagonal boron nitride, where positively charged boron atoms and negatively charged nitrogen atoms on each plane combine with charged atoms on the next plane. Both graphite and hexagonal boron nitride can be used as lubricants. However, hexagonal boron nitride atoms have a certain polarity between them, and their conductivity and thermal conductivity are weaker than those of graphite.
+1 and+2 oxidized boron:
Although boron almost exists in the+3 oxidation state on Earth, there are also compounds with an oxidation state less than 3. For borohydride and borides, their oxidation state usually has little practical significance. However, halogens can form derivatives with boron in the+1 and+2 oxidation states. Boron monofluoride, which is an isoelectronic body with nitrogen gas, generally exists in a polymerized form and cannot be separated in a concentrated form. However, certain compounds such as diboron tetrafluoride and boron tetrachloride have been determined in their structures.
Organic boron chemistry:
Organic boron compounds are a class of organic derivatives of boron, such as trialkyl boron. In organic chemistry, organic boron compounds are widely used due to their various changes, with the most common use being borohydride reactions. There is a lot of content in organic boron chemistry, and due to the word limit of the column, I can only briefly introduce these.
Synthesis of organic borides:
Synthesized by Grignard reaction:
Simple organic boron compounds, such as triethyl boron compounds or tri (perfluorophenyl) boron compounds, can be prepared by reacting with Grignard reagent using * * * (usually a complex of ether) or ethyl or perfluorophenyl boron compounds.
From olefin synthesis:
Boron can react quickly with olefins after combustion, which is called borohydride reaction. Ethylboron is usually present as a dimer, while methylboron can form a 1:1 complex with alkaline solvents such as tetrahydrofuran. In simple electrophilic addition reactions involving hydrogen halides, the Martensian rule is followed, and the same applies to the electrophilic addition of boron to double bonds. Due to the weaker electronegativity of boron compared to hydrogen, hydrogen atoms are always added to the most substituted carbon atoms. The positive charges formed on olefins are added to the carbon atoms with the most substituents, while some negatively charged hydrogen atoms are added to them. The remaining boron atoms are added to the carbon atoms with fewer substituents. When boron atoms are replaced by hydroxyl groups, the addition of water to double bonds follows the anti Markov rule.
When boron compounds have large steric hindrance substituents, their chemical properties are very special. An organic boron compound commonly used in organic synthesis is 9-BBN (9-borobicyclic [3.3.1] nonane). This compound can be prepared by the complete reaction of cyclooctadiene with ethylboron. In the process of cis borohydride, stereospecificity occurs, which involves addition reactions on the same side of the olefin. In this synergistic reaction, the transition state is represented as a square composed of carbon atoms, hydrogen atoms, and boron atoms. In this square, there is the maximum electron cloud overlap between the two double bonds, p-orbitals, and the empty orbitals of boron atoms.
Synthesis by boron pyrosynthesis:
The C-H bond boronization reaction is an organic reaction catalyzed by transition metals, which produces organic boron compounds through the boronization of C-H bonds between aliphatic and aromatic hydrocarbons. The common reagent in this reaction is the double linked pinacol borate.
Metal catalyzed C-H bond boron pyrocompletion reaction:
Using Cp * Rh( η ₆ Me ₆) can be used as a catalyst for highly selective boron pyrocompletion of the primary C-H bond. It is worth noting that even in the presence of heteroatoms in the carbon hydrogen chain, the selection of the primary C-H bond has an exclusive effect. The borylation of C-H bonds catalyzed by rhodium salts can occur selectively without relying on the position of heteroatoms. A series of acetals, ethers, amines, and alkyl fluorides can selectively undergo boron pyrocompletion at low steric hindrance and electron withdrawing primary C-H bonds. In addition, in the absence of primary C-H bonds, such as when cyclohexane is the substrate, no reaction will occur.
The selective functionalization of primary alkane bonds is based on the formation of secondary alkane metal complexes, resulting in the formation of more favorable primary alkane metal complexes in terms of kinetics and thermodynamics.
The high stability of primary and secondary alkyl complexes can be attributed to several factors. Firstly, the primary alkyl complex is spatially superior to the secondary alkyl complex. Secondly, some negative charges typically exist in metal alkyl complexes α- On carbon, and the primary alkyl ligand can better support some negative charges than the secondary alkyl ligand. The use of a mechanical detection called hydrogen deuterium exchange can investigate the origin of selectivity for the pyrocompletion of aliphatic C-H boron using rhodium catalysts. Hydrogen deuterium exchange shows that the regional selectivity of the entire process shown below is due to the selective cleavage on the primary C-H bond and the selective functionalization of metal alkyl intermediates on secondary metal alkyl intermediates.
The synthesis effect of aliphatic C-H borylation has been applied to modify polymers through boron pyrophosphorylation, and then oxidize them to form hydroxyl functional group polymers.
Aromatic C-H boron pyrocompletion:
For symmetrically substituted 1,2- and 1,4-substituted aromatic hydrocarbons, the addition of pinacol borate only occurs in one position. Symmetric or asymmetric 1,3-substituted aromatic hydrocarbons are selectively boronized, as only one C-H bond is spatially acceptable.
This is the opposite of electrophilic aromatic substitution, where the regioselectivity is controlled by electronic effects.
The synthesis of aromatic C-H boron pyrocompletion is as follows, where 1,3-disubstituted aromatic compounds can be directly converted into 1,3,5-organic boron pyrocompletion compounds and subsequently functionalized.
The functionalization of aromatic C-H was successfully incorporated into the synthesis of a dimeric alkaloid (Compenadine A), which is a alkaloid of Lycopterygii and can enhance the mRNA expression of nerve growth factor (NGF) and the production of NGF in human glial cells. Promoting the growth of neural networks is of great significance in the treatment of diseases such as Alzheimer's disease. The reaction starts by directly boronizing the C-H bond on the aromatic group, followed by a Suzuki coupling reaction, and finally cutting the tert butoxycarbonyl protecting group to successfully synthesize Complex A.
Heteroaromatic boron pyrogenation:
Heteroaromatic hydrocarbons can also undergo boron pyrogenation under iridium catalytic conditions. However, in this case, the selection of sites is controlled by electronic effects, where furan, pyrrole, and thiophene are α The C-H bond at the position reacts with the heteroatom. In this case, it is best to react by interacting with the heteroatoms α The C-H bond at the position reacts because it is the most proton withdrawing C-H bond with good reactivity.
Directional ortho boron completion:
Using the same catalyst can achieve regioselectivity by guiding groups without the need for substituents as spatial media. For example, the dimethyl-hydrosilyl group on aromatic hydrocarbons undergoes iridium catalyzed boron pyrocompletion at the adjacent C-H bond, which can be directed towards the silane group. In the case of the use of hydrogenated silane based directing groups, the ortho selectivity is due to the reversible addition of Si-H bonds to the metal center, resulting in the preferential cleavage of C-H bonds by the ortho hydrogenated silane substituents.
Aromatic boron pyrogenation mechanism:
The iridium complex with triboron pyrocompletion can promote these reactions, leading to the completion of C-H boron pyrocompletion of aromatic and heteroaromatic hydrocarbons. Kinetic studies and isotope labeling indicate that the iridium complex with three boronic groups reacts with aromatic hydrocarbons during the catalytic process. The following figure is a form of catalytic cycle that can be used for ortho substitution of hydrogen containing silane compounds. The kinetic data shows that the coordination between the tricyclic complex and cyclooctene rapidly and reversibly dissociates cyclooctene, forming a 16 electron triple helix complex. In the case of using benzyldimethylsilane as the guiding group, benzyldimethylsilane reacts with iridium trithiophene catalyst by reversibly adding Si-H bonds to the metal center, and then selectively activates the adjacent C-H bonds through oxidative addition and reduction elimination reactions.
Variable selective boron completion: Variable selective boron completion is an important synthetic conversion. However, this steric separation is completely spatially oriented and limited to 1,3-disubstituted aromatic compounds. Using the same matrix in the reaction can alter the selectivity of another ligand position. The variable selective boron pyrophosphorylation is driven by two effects, one is electrostatic interaction, and the other is the interaction of secondary nitrogen boron bonds.
Secondary variable selective boron pyrocompletion can better describe the boronization interactions between various carbonyl compounds.
Reduction reaction:
Chiral amino alcohols and boron can asymmetric reduce prochiral ketones to obtain corresponding secondary alcohols containing 60% enantiomers. Chiral amino alcohols will react with boron to form aldehyde oxy amine boron coordination compounds. This complex is considered to contain a relatively rigid five membered ring system, which has thermal stability and hydrolysis stability, and is soluble in various proton and non proton solvents.
Boron flame retardant and chiral amino alcohol form oxazole boron flame retardant. In addition, oxazolboron can catalyze prochiral ketones and undergo rapid enantioselective reduction in the presence of tetrahydrofuran methyl boron. This reaction is called the Kori Baksh Chaitian reduction reaction or CBS reduction.
from α- 3-Boron obtained from the reaction of pinene with 9-BBN- α- Pinene-9 boron bicyclic [3,3,1] nonane, which can quickly convert α- D-benzaldehyde is reduced to (S) - (+)- α- D benzyl alcohol can complete directional asymmetric induction reactions.
3-Boron- α- Pinene-9 boron bicyclic [3,3,1] nonane can be used as a reducing agent and is referred to as pinene boron pyrogenation. The reaction of asymmetric carbonyl reduction using the enantiomer of pinene boron pyrogenation is referred to as Mitterrand pinene boron pyrogenation reduction.
Active pyranone can be synthesized through this stereoselective method of reaction. Three chiral centers can be formed at C-4 ', C-5, and C-1' through Mitterrand reduction and Sharpley asymmetric dihydroxylation.
Coupling reaction:
The improved Mannich reaction can effectively synthesize allylamine. In this improved Mannich reaction, they found that vinyl boric acid can participate as a nucleophilic reagent in the production of pure * * *. This improved Mannich reaction is called the Patassis boric acid Mannich reaction.
The use of tartaric acid modified allyl borate esters can effectively control the reaction of chiral and non chiral aldehydes. In addition to allyl alcohols, borate esters of 2-butene-1,4-diol and trans ortho-diol can also act on chiral synthesis. This reaction is called Rush asymmetric allylation.
The use of non enantioselective Rush asymmetric allylation as a key reaction can introduce two chiral intermediates and complete (8S, 11R, 12R) - and (8R, 11R, 12R) - B2 endorphins in eight steps. These two chiral intermediates can also synthesize two other non enantiomers.
Arylated (E) - olefins can be efficiently synthesized by the reaction of aryl halides and alkyl-1-enylboron under the catalysis of tetra (triphenylphosphine) palladium and alkali. This reaction can also be extended to other organic boron compounds and other alkenyl, aryl, alkyl halides, trifluoromethanesulfonic acid esters, and other substances. These palladium catalyzed coupling reactions between organic boron compounds and these organic halides to form carbon carbon bonds are called Suzuki reactions.
The Suzuki reaction can synthesize protein precursors from certain organisms. The structure of the fungal toxin Arturate III in the genus Alternaria can be determined through nuclear magnetic resonance spectroscopy analysis. In the synthesis method, the Suzuki coupling reaction was used in conjunction with highly functionalized borate esters and butenolactones to produce the precursor of altulinic acid III.
Copper powder can catalyze the reaction of aryl halides with phenols to form diaryl ethers. This reaction is called the Ullmann condensation reaction. Afterwards, this reaction can be extended to synthesize aromatic amines with aryl halides and amides in the presence of potassium carbonate and cuprous iodide. This reaction is called the Kobayashi reaction. Afterwards, organic potassium trifluoroborate is used to react with aliphatic alcohols, aliphatic amines, or aniline to synthesize aryl ethers or aromatic amines.
Organic boride chemical reaction:
Borohydride oxidation:
In organic synthesis, borohydride reaction is commonly used to continue the conversion to other functional groups by introducing boron groups. The borohydride oxidation reaction provides a method for synthesizing alcohols by first reacting with boron and then oxidizing with hydrogen peroxide. During this reaction, if the oxidant is replaced with chromium oxide, the hydroxyl group will continue to oxidize into carbonyl compounds.
The reaction between organic borate and boron involves transferring the nucleophilic groups attached to boron to intermolecular or intramolecular electrophilic centers. α,β- Unsaturated borate and its application in α Borates with departing groups are highly sensitive to the transfer of electrophilic groups, which mainly involve the transfer of electrophilic groups α Intramolecular 1,2 migration of sites. The obtained organic boron can undergo oxidation or proton decomposition after combustion, resulting in various organic products, including alcohols, carbonyl compounds, olefins, and halides.
Main introduction:
Organic boron (R Å B) and borate (R ₄ B ⁻, generated by R ⁻ addition to R Å B) have boron carbon bonds, with polarity oriented towards carbon. Therefore, the carbon connected to boron is nucleophilic, and in borate salts, this property can be used to transfer one of the R groups to the electrophilic center within the molecule. In this case, the nucleophilic R group undergoes 1,2- migration to the electrophilic carbon connected to boron. Then, the obtained recombinant boron can be completely oxidized by fire or undergo proton decomposition to obtain organic products. The specific introduction is shown in the following figure.
The borohydride of olefins or alkynes is an effective method for producing boron. However, the use of boron flame retardant or boron flame retardant analogues can result in only 33% of the original olefins being converted into products after oxidation or proton decomposition, and the remaining olefins producing additional boron containing by-products. Using 9-BBN as a hydrogenation reagent can solve this problem.
Mechanism and Stereochemistry:
Main mechanisms:
Boron alone usually does not have sufficient nucleophilicity to transfer alkyl groups to electrophilic centers. However, after the nucleophilic attack, the obtained borate is highly nucleophilic. If the nucleophilic reagent is present α If the position contains unsaturated functional groups or leaving groups, one of the R groups connected to boron can migrate to electrophilic α On carbon, please refer to the following figure for details. The tendency of organic groups to migrate depends on their ability to stabilize negative charges: the general rule is alkynyl>aryl ≈ alkenyl>primary alkyl>secondary alkyl>tertiary alkyl. When migration occurs, the configuration of the migrated carbon remains unchanged, and when it reaches the endpoint, the configuration undergoes a reversal, but the configuration at the endpoint is SP ³ Hybridization. Double (de icing based) borohydride and 9-BBN are commonly used as "virtual" borohydride reagents, as only the R group on borohydride olefins is possible for nucleophilic activation and migration.
In this situation
Technological innovation
Honesty is the foundation
Contact Number: +86-15698999555 |
Address: NO.6 ,SHENGHUA STREET,TAIHE DISTRICT, JINZHOU CITY, LIAONING PROVINCE, CHINA. |